Freshwater Aquarium Water Quality: The Nitrogen Cycle & Optimal Water Chemistry
Maintaining a healthy freshwater aquarium takes a little time and effort, but the rewards are well worth it for you and your fish! Spending an average of 30 minutes to an hour on your freshwater aquarium water quality each week will ensure years of enjoyment and help avert most major problems.
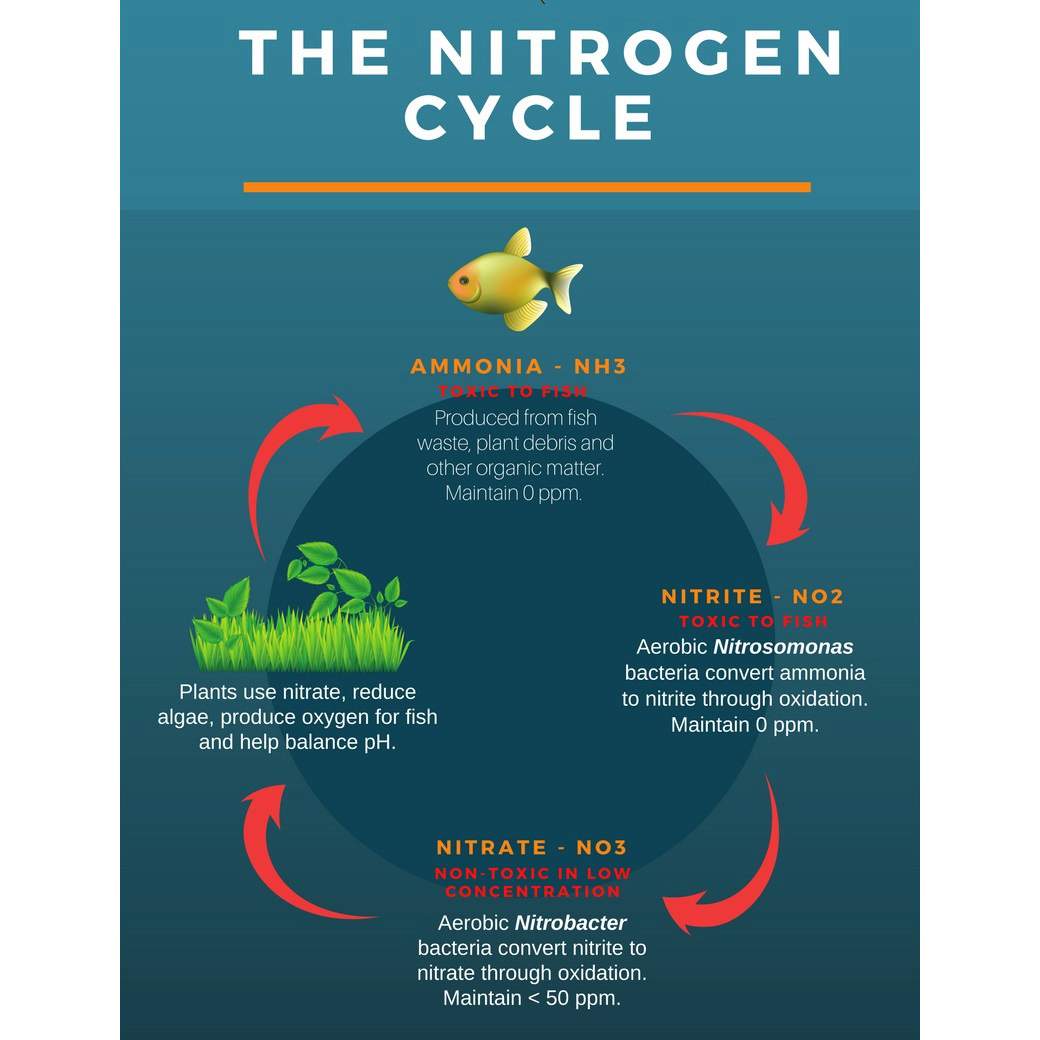
The Nitrogen Cycle
The aquarium nitrogen cycle is the mechanism by which waste is processed in an aquarium. Freshwater aquarium fish release waste in the form of ammonia (NH₃), which is toxic to all aquatic organisms. Nitrifying bacteria living in the filter, gravel bed, and on solid objects in the aquarium convert ammonia to nitrite (NO₂) which is also toxic. Nitrite is then converted to nitrate (NO₃) by a different set of nitrifying bacteria. Nitrates are not toxic to fish per se, however, long term exposure to high levels can stress them, stunt growth, damage organs and make them more susceptible to disease. Nitrates are used by aquatic plants but if allowed to accumulate nitrates contribute to unsightly algae growth. Other sources of biological waste include uneaten food and dead plant material.
Seeing cloudy water? Take a look at this helpful article on how to help!
Establishing nitrifying bacteria, or “cycling” a new freshwater aquarium begins after you add the first fish and can take several weeks to even months in larger aquariums. Simply allowing your aquarium to run for 24 to 48 hours after first setting it up is not cycling. Adding too many fish at once or overfeeding a new freshwater aquarium will cause ammonia and nitrite to rise to unsafe levels – a condition known as “New Tank Syndrome” – and may result in fish loss. It can also prolong the cycling process. Seeding a new aquarium with a small amount of gravel or biological filter media from an established, healthy aquarium will help speed up the cycling process. Stock your new aquarium gradually, feed sparingly and test water regularly until ammonia and nitrite levels stabilize at zero.
Take a look at our 3 Tips to Succeed Video for more helpful tips.
Water Chemistry and Testing to Maintain Freshwater Aquarium Water Quality
You don’t have to be a biologist or chemist to be a successful aquarist but understanding the basics of water chemistry and water quality will help you maintain a healthy, trouble-free aquarium. Testing your freshwater aquarium water quality helps avoid problems when it is first set up, and ensures healthy, vibrant fish over the long term. Below, we outline some water quality parameters that affect your fish.
Testing Aquarium pH
pH – the measure of whether water is acidic (pH 1 to 7.0) or basic (pH 7.1 to 14). 7.0 is considered neutral. Most freshwater aquarium tropical fish do best at a pH of 6.8 to 7.8, although certain fish may require higher or lower levels. The pH of an aquarium tends to drop over time due to the breakdown of organic material, and the best way to prevent this is through regular partial water changes.
Testing Aquarium General Hardness
GH (General Hardness) – the measure of calcium, magnesium and other ions in water, sometimes referred to as Total Hardness. Most freshwater fish adapt to a wide range of general hardness, and it is best to adjust them to your local water. Unless you are trying to keep or breed fish from areas of the world with extremes in water hardness, do not try to manipulate the pH or hardness level in your aquarium with additives -- it can send your fish on a water chemistry roller-coaster ride that severely stresses them. If your tap water is excessively hard or soft, the use of reverse osmosis or deionized water with added Aqueon Water Renewal, Kent Marine RO Right or Liquid RO Right, is a more natural and stable way to adjust GH.
Testing Aquarium Carbonate Hardness or Alkalinity
KH (Carbonate Hardness or Alkalinity) – the measure of carbonates and bicarbonates in water; the ability to resist changes in pH. Carbonates help stabilize pH in the aquarium and are replenished through regular partial water changes. Hobbyists should be cautious when researching fish they intend to keep, as many fish that have origins in tropical rainforests where water is extremely soft and acidic, are captive bred nowadays in water with much higher alkalinity and pH levels. Attempting to duplicate conditions in the wild may stress captive bred fish.
Testing Aquarium for Ammonia
Ammonia (NH₃ and NH₄+) – the primary component of fish waste. Ammonia is harmful to fish and tends to rise in newly established aquaria due to a lack of nitrifying bacteria, adding too many fish at once, overfeeding or a combination of these factors. It can also rise in heavily populated tanks due to generous feeding and/or insufficient filtration. In water, ammonia exists simultaneously as free ammonia (NH₃) and ammonium ion (NH₄+). Aquarium water test kits usually measure NH₃ and NH₄+ combined. Ammonia is more toxic at higher temperatures and pH levels above 7.0, and less harmful at lower temperatures and pH levels below 7.0. The only safe ammonia level is zero.
Testing Aquarium for Nitrite
Nitrite (NO₂) – nitrifying bacteria living in the filter and aquarium convert ammonia to nitrite (NO₂). A rise in nitrite usually follows an ammonia spike. Nitrite inhibits oxygen exchange to fishes’ bloodstream, thereby causing them to suffocate. Fish that are experiencing nitrite toxicity will often breathe rapidly and gasp at the surface (although there are other reasons they do this), and their gill filaments will turn from bright red to dull brown or grey in color. The only safe nitrite level is zero.
Testing Aquarium for Nitrate
Nitrate (NO₃) – nitrite is converted to nitrate (NO₃) by nitrifying bacteria. While nitrate is far less toxic to fish and other aquatic organisms than ammonia and nitrite, it can stunt your fish’s growth and long-term exposure to high levels stresses them and compromises their immune systems. Nitrate is also a major contributor to algae growth. Some tap water sources are high in nitrate, so it is a good idea to test your tap water before using it. Nitrate toxicity to fish varies depending on species, age and overall health, but levels above 50 ppm are undesirable in freshwater aquaria. Regular partial water changes, proper filtration, sensible stocking and feeding habits as well as the use of live plants will help keep nitrates in check.
Testing Aquariums Phosphate
Testing Aquarium for Phosphate
Phosphate (PO4) – common sources for phosphate in an aquarium include tap water, fish food, certain types of activated carbon, decaying organic material, and certain types of buffers and water additives. While phosphate is not particularly harmful to fish, it is a major contributor to algae blooms, especially “hair algae”. To maintain low phosphate in an aquarium, do regular partial water changes using phosphate-free water, feed sparingly using low phosphate containing foods, keep the aquarium free of organic debris, and use Kent Marine Reef Carbon and Phosphate Sponge in the filter.
Freshwater Aquarium Optimum Water Conditions Chart




